Neonatal Biotinidase
A fluorometric assay designed for precise determination of biotinidase enzyme activity from dried blood spot samples — enabling early detection of biotinidase deficiency in newborns.
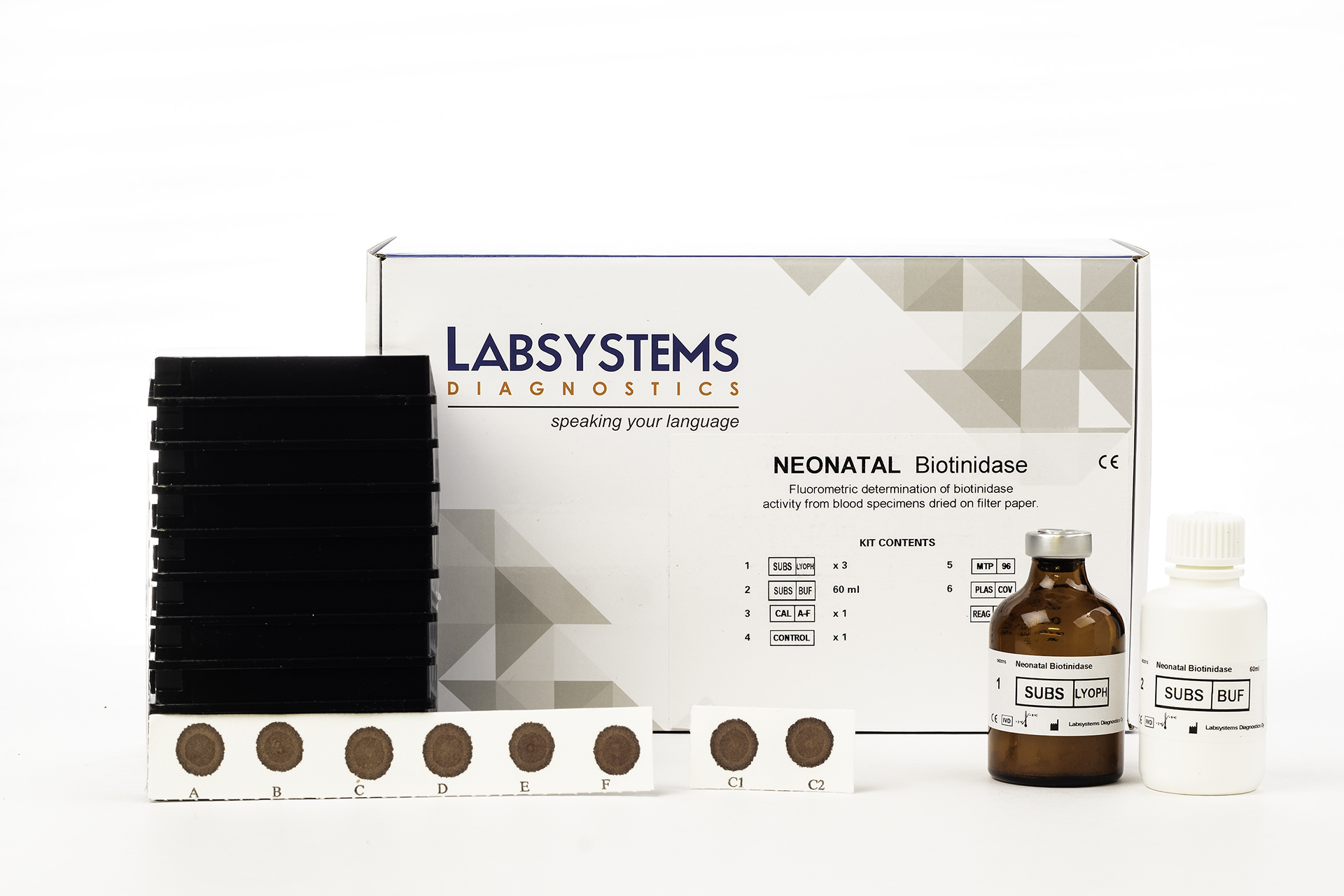
Overview
The Labsystems Diagnostics Neonatal Biotinidase Kit offers an enzymatic method to measure biotinidase activity using dried blood spot samples. The test utilizes fluorometric detection, producing a fluorescent reaction product that directly reflects enzyme activity. This direct assay approach ensures accuracy independent of dietary biotin or protein intake, supporting consistent and reliable screening outcomes in newborn populations.

Features
- Direct enzymatic assay — unaffected by dietary biotin or protein intake
- No centrifugation or filtration steps required
- Fluorometric detection ensures high sensitivity and precision
- Compatible with Labsystems Diagnostics NS2400 automated system
- Ready-to-use solid plates simplify workflow and reduce handling errors
Technical Specifications
- Format : 10 solid plates (96 wells each)
- Total Wells : 960
- Catalogue Number : 6199821
- Detection Method : Fluorescence
- Sample Type : Dried blood spot
IS Modules
The Neonatal Biotinidase assay seamlessly integrates with Labsystems Diagnostics NS2400 automation modules, offering:
- Automated plate handling and result calculation
- Configurable test setup and analysis workflows
- Secure data management with supervisor and user-level access
- Enhanced throughput for high-volume screening laboratories
Inquiry Form
